Could cancer immunotherapy drugs one day be taken orally? A new study from Fudan University provides a potential technological foundation.
On March 19, teams led by LU Boxun from Fudan University and SONG Haikun from Greater Bay Area Institute of Precision Medicine (GBA-IPM), together with SHENG Chunquan at Naval Medical University and other collaborators, published a paper in Cell titled “Hijacking ER-associated degradation (ERAD) for targeted degradation of transmembrane proteins.”

By exploiting the requirement of transmembrane proteins to fold in the endoplasmic reticulum (ER), they harnessed the ERAD pathway to create a new strategy of targeted protein degradation—ERAD-engaging chimera(ERADEC).
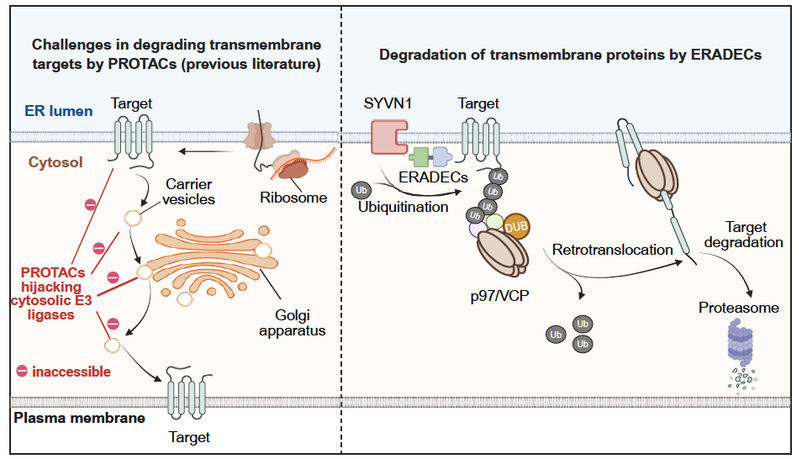
This approach enables efficient, selective degradation of transmembrane proteins using small molecules, potentially advancing oral cancer immunotherapy and expanding drug discovery for membrane-protein-related diseases.
Cells can be compared to highly organized factories, where proteins, their essential products, are synthesized, folded, transported, and degraded every day. Any error in this process could ultimately lead to disease.
Cells maintain quality via the endoplasmic reticulum (ER), where ERAD detects misfolded proteins, tags them with ubiquitin, and directs them to the proteasome for degradation.
Traditionally, ERAD was thought to only eliminate defective proteins. However, the researchers proposed a bold idea: Can we deliberately redirect disease-causing proteins into this system and allow the cell to destroy them?The idea led to a new technological breakthrough in targeted protein degradation—a field focused on designing molecules that selectively eliminate harmful proteins rather than merely blocking them.
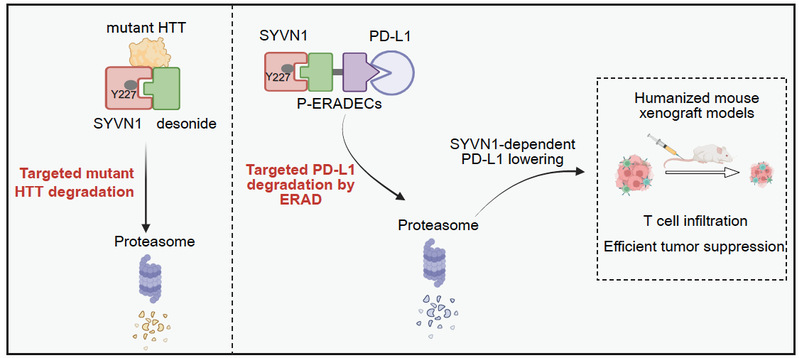
The key challenge was identifying a molecule capable of engaging the ERAD system. The team discovered that desonide can bind the ER E3 ligase SYVN1 and promote degradation of a disease protein. Based on this, they proposed ERADEC—a bridge-like molecule with one end that binds SYVN1, the other that binds the target protein, and a linker connecting the two. This forms a ternary complex, triggering ubiquitination and degradation.

Using PD-L1 as a model target, they showed that ERADEC significantly reduces its protein level in a SYVN1- and ERAD-dependent manner. In mouse tumor models, ERADEC demonstrated strong anti-tumor effects, even outperforming existing antibody therapies. AI tools were also used to predict protein structures and binding modes, aiding molecular design.

Although current compounds are not yet orally available, improved versions show partial oral bio-availability. The team has already licensed related patents and is currently developing next-generation compounds to put this discovery into clinical practices.
Original paper:
https://www.cell.com/cell/abstract/S0092-8674(26)00105-4
(END)
Writer: ZHANG Xinning
Proofreader: YANG Xinrui
Editor: WANG Mengqi, LI Yijie


